Ansel Lim is currently in the fourth year of medical school and, under the guidance of his supervisors Dr Jabed Iqbal and Dr Joe Yeong, has been carrying out research into triple negative breast cancer at the Department of Anatomical Pathology at the Singapore General Hospital.
We caught up with him about the winning image, his work and the next steps for his research.
Tell us about your image and the research behind it
I have been studying the role of a specific type of immune cell, known as a tumour-associated macrophage, which has a surprising role in triple negative breast cancer. These macrophages are thought to be involved in actually preventing the immune system from attacking tumour cells and may drive the development of secondary breast cancer. Our aim is to find out whether we can predict survival outcomes for patients by counting the number of these immune cells present in a triple negative tumour.
In my image, we can see a tumour-associated macrophage in the centre (coloured red and yellow) mingling closely with other macrophages and immune cells known as lymphocytes and surrounded by tumour cells (coloured blue).
The image comes from a tumour sample taken from just one of around 300 patients in our cohort of triple-negative breast cancers. We used a technique called quantitative multiplex immunohistochemistry – which is quite a mouthful to say – to look at each and every one of these samples and find out how many macrophages there are in each sample. We will then run sophisticated statistical tests to work out if there is a relationship between the number of macrophages present and the patient’s chances of survival.
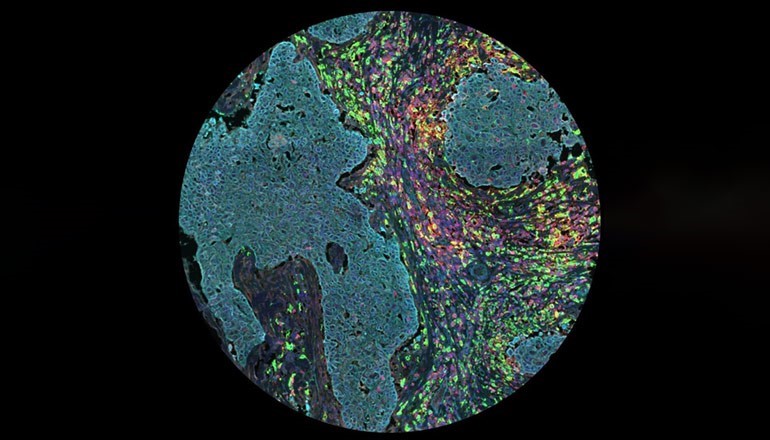
Ansel's winning image
What is quantitative multiplex immunohistochemistry?
This technique builds on a well-established process called immunohistochemistry, which uses specialised fluorescent markers to identify and study the different proteins produced by a cell. In this image we used these fluorescent markers to look for seven specific proteins; the tumour-associated macrophage is highlighted in red and yellow due to the presence of two of these proteins, CD68 and CD163, which are found on the surface of the cell.
Quantitative multiplex immunochemistry, or computer-aided immunochemistry, is the next generation of immunohistochemistry. The technique allows researchers to look at multiple proteins simultaneously (a limitation of conventional immunohistochemistry), and allows us to count the number of cells that produce a particular protein with more precision. What this means in practice is that we can identify the different proteins that are produced by different patients’ tumours with much greater accuracy.
How does your research have the potential to benefit patients?
We are currently seeing huge advances being made in the development of immunotherapies for diseases such as melanoma and non-small cell lung cancer. We want to see whether immunotherapies are a viable option for triple negative breast cancer, which is a particularly aggressive form of the disease. In the spirit of personalised medicine, we believe that treatments should only be used if the patient is likely to benefit from them, so it’s essential that we can confirm that the targets of these drugs are present on the patient’s cancer and immune cells.
We also believe it is crucial to find out how the immune cells that surround and infiltrate the tumour (known as the microenvironment) affect survival, long-term outcomes, and possibly response to immunotherapy. We are therefore studying the immune microenvironment in the hope that we can find relationships between what a tumour looks like under the microscope, and how a tumour behaves in the patient, including of course how it responds to therapy.
In comparison with other conditions, such as melanoma, it’s still very early days for the use of immunotherapies to treat triple-negative breast cancer. Nevertheless, pathology research such as ours may one day help our clinical colleagues to rationally select the patients who are most likely to benefit from immunotherapy.
What are the next steps for you and your research?
I have recently found an exciting relationship between a subpopulation of macrophages and overall survival and so in the coming weeks, I will be validating these results. Research never ends, the questions never stop coming, and the answers get closer every time, but new questions constantly emerge.
Once I have completed my medical degree, I intend to train in anatomical pathology, after which I would like to pursue formal research training and complete a doctoral degree.